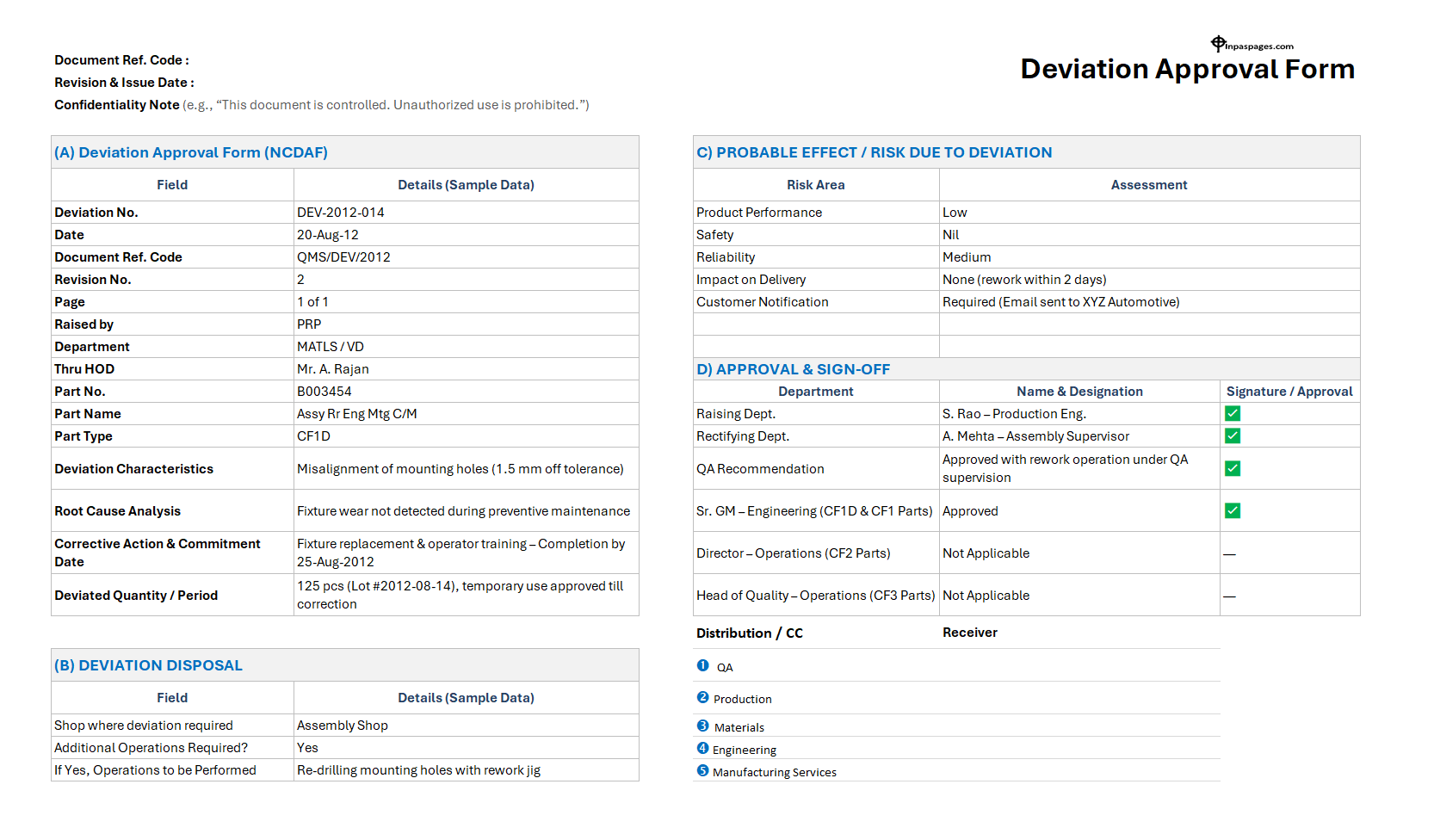
A deviation approval form is an official quality control document. It is used to formally approve, and control, deviations.
Approve what?
To formally approve deviations from established standards, procedures, or specifications to ensure quality and compliance.
Formally approved specifications in industries.
Manufactured and regulated: A specific scope from standard operating procedures (SOPs), drawings, process parameters, material requirements, or regulatory conditions.
What does this form ensure?
- Deviations are scientifically and technically justified.
- Evaluated for quality, safety, and compliance risks.
- Approved by authorized personnel.
- Implemented under controlled conditions.
What is the purpose of this approval form?
Quality Assurance and Control
In manufacturing sectors, it often happens that normal standards cannot be followed. In such cases, the purpose is to ensure that the quality of the product is not compromised.
Control Deviations
It is necessary to ensure that deviations from standard operating procedures (SOPs), work instructions, specifications, drawings, etc. are exceptional.
Ensure product quality and safety
This form helps to understand whether the deviation affects the quality, or customer requirements. It is necessary to ensure whether it poses a safety or regulatory risk. Deviations that pose a risk to quality or safety are not approved.
Maintain compliance with standards
If the manufacturing unit follows a quality management system, then it must comply with its rules and its procedures. Deviation approval forms are considered necessary for such systems.
Risk Assessment and Impact Analysis
The root causes are identified before issuing this approval form.
The risk that is inherent in quality, delivery, safety or other compliance is assessed. This process also prevents uncontrolled risks from entering.
What is the importance of approval forms for international standards?
| ISO 9001 perspective | IATF 16949 perspective | GMP perspective |
| To control any type of change. | Customer-specific requirements must be strictly observed. | About formal deviation reporting. |
| The concept of this form is risk-based. | About permitted deviations for product and process changes. | Investigation and impact assessment are required.
|
| Control of unsuitable products or outputs. | Required for traceability and documented approvals. | Approval of the quality assurance team is required before use or release. |
When is a deviation approval required?
- When it is found that non-conforming or alternative raw materials are being used at the workplace, the need for deviation approval arises.
- Whenever there is a need to temporarily relax the process parameters.
- Perhaps there is a breakdown in the running equipment, and at that time, an approval is required.
- Deviations regarding tooling or fixtures are not used without approval.
- In situations where variations in environmental conditions arise.
Apart from that, approval is required in deviations such as supplier-related deviations, emergency production to meet delivery commitments, etc. Also, the deviation approval team ensures that the risks remain under control.
Whose roles and responsibilities are?
| Production Team | This team identifies deviations. And ensures that the approved conditions are being followed. |
| Engineering | This team evaluates the technical feasibility and impact. |
| Quality Assurance
| This is an important responsibility. Here, international standards such as ISO, IATF and GMP that ensure compliance with the requirements of quality management systems are follows. |
| Management
| When deviations are serious, such as high-risk or customer-impacting deviations. In such cases, approval by management is require. |
Deviation Approval Form