Corrective action Preventive action is an action taken to eliminate a defect or problem, as well as the cause of the problem. Corrective action is the process of eliminating an existing nonconformity, defect or problem, while preventive action is taken to eliminate the cause of a potential nonconformity or problem. The only difference between the two is that corrective action is reactive in nature while preventive action is proactive in nature.
Corrective action is an action taken to eliminate a defect or undesirable situation in a production process, product or system if it has occurred, while preventive action is an action taken to eliminate the cause of a possible defect or undesirable situation in a production process, product or system before it occurs.
How to identify?
Both these methods are taken to prevent the recurrence of defects. Therefore, the following characteristics should be kept in mind to identify both of them.
| Corrective Action (CA) | Preventive Action (PA) |
| Reactive action is taken as a measure. | It is proactive in nature, which is taken to eliminate the causes of the defect before it occurs. |
| It is started after the problem is detected. | When it is implemented before the problem occurs. |
| In this, steps are taken to eliminate the root cause of the problem. | In these processes, steps are taken based on identifying the risk and analyzing it. |
| Due to this, the recurrence of a similar problem is prevented. | While this prevents problems that may occur in the future. |
These processes are very necessary and useful. Corrective action identifies and eliminates the causes of past incidents so that the same defect does not reoccur, while preventive action focuses on the analysis of where defects are likely to occur in the future and eliminates them in a timely manner so that they do not occur.
Why is CAPA important?
CAPA is a process in which all the processes, resources, and systems in the production area are identified and eliminated where defects are found or where there is a possibility of defects. Here the important roles are mentioned as follows:
Quality Improvement
When the CAPA process is implemented, it is natural that the defects or systemic weaknesses in the processes, resources, and systems are identified and eliminated. So that the product continues to improve. Which improves the quality of the product.
Compliance with standards
Whenever a manufacturer adopts international or any other standards or criteria, it has to comply with its standards. Such as ISO 9001, Lean Manufacturing or others. In which organizations control non-conformities and risks by using corrective and preventive approaches.
Cost Reduction
If the defects in the production processes are removed on time, it is natural that once the production processes are completed, it will produce goods with quality. In which, incorrect rework, waste or wastage will be reduced, downtime of machinery and resources will be prevented. Overall, costs will be reduced.
Customer Satisfaction
If quality products are delivered on time along with goods, it is natural that customer satisfaction will increase. The credibility of the manufacturer in the market will increase.
Risk Management
When we carry out CAPA, every preventive measure has to be taken. So that potential risks in the production areas are focused on and eliminated before they increase. Due to which risk management is maintained in the production areas.
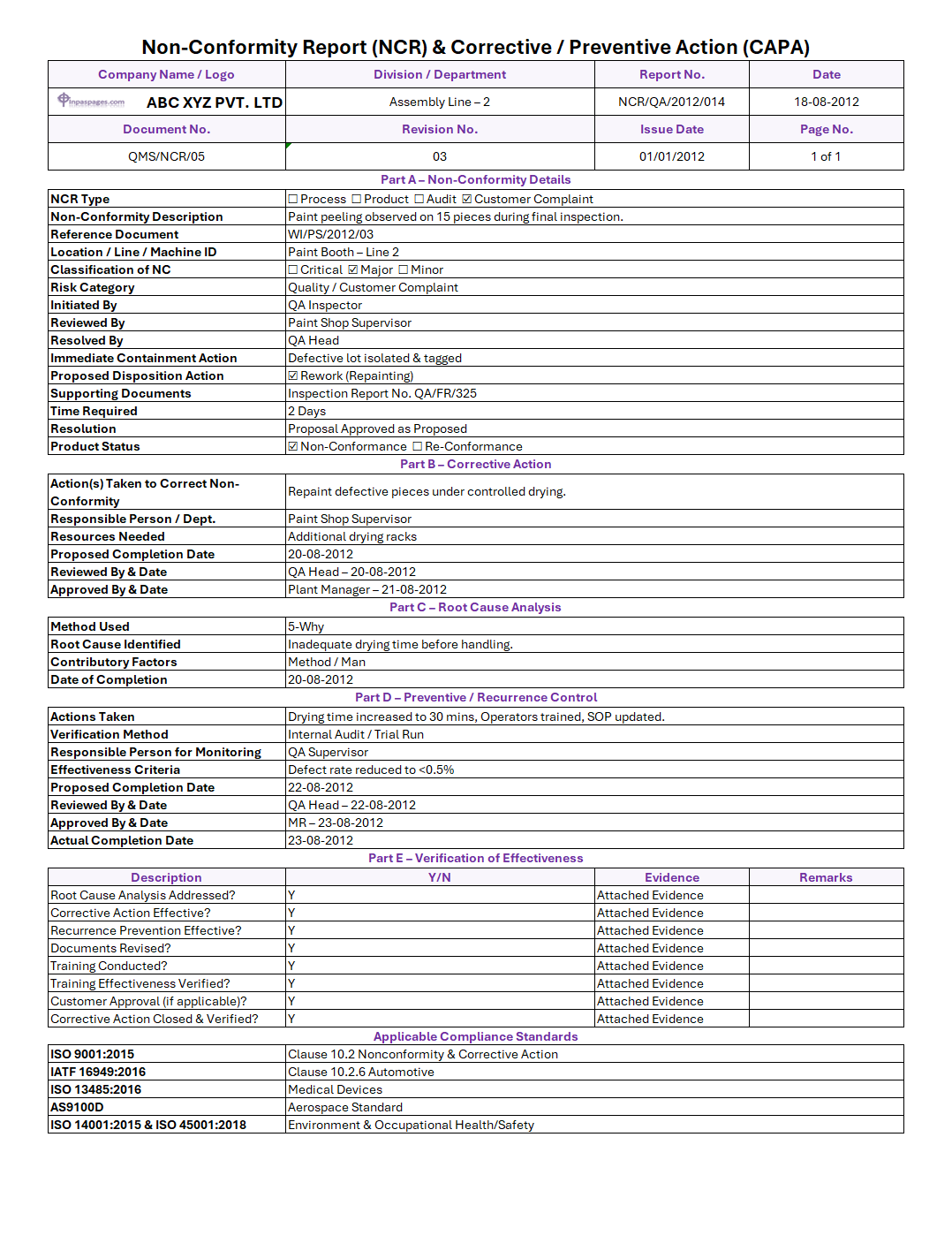
Non-Conformity corrective action report
When an inspection or audit is conducted in a manufacturing process, if a defect or non-conformity is found, it is considered a non-conformity. Along with this non-conformity action report, CAPA processes are also required to eliminate the non-conformity of those processes. So that both these reports can be created together.
A non-conformity corrective action report is a record of defects or non-conformities found after an inspection or audit, which is used to identify, analyze, correct and eliminate the root cause of the non-conformity in quality, safety and management systems so that it does not happen again.