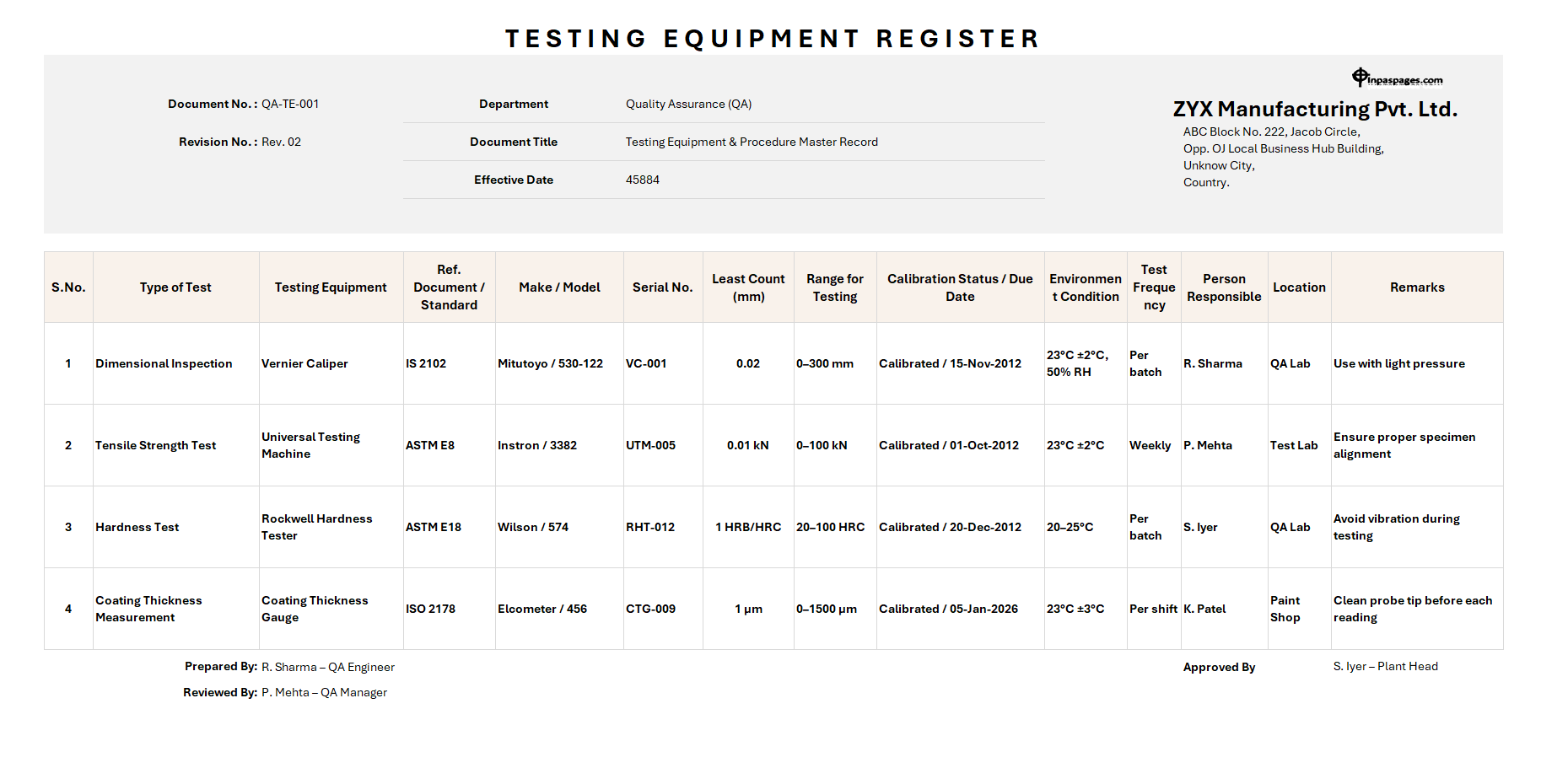
Testing equipment register
A testing equipment register is recording sheet that may helpful to describing and tracking all measuring, inspection, and testing equipment.
This equipment, and instruments are generally useful for verifying product quality, process accuracy as per standards. In the register, all the important information such as equipment identification number, specifications, calibration status, location, department, as well as maintenance history.
The key purpose of this register is to maintaining records of the equipment, instrument, and devices that use in the measurement and calibration purposes.
It helps to ensure traceability, accuracy, and effective control of measurement systems. The quality team can ensure that the equipment is used for the testing purpose are accurate, and calibrated as per standards.
The register is covering every testing or measuring instrument that use for quality inspections. The information is for calibration planning, verification tasks, Preventive maintenance, equipment traceability, standards and compliance, etc.
Examples of Testing equipment, instrument, and devices are weighing balances, temperature sensors, vernier, micrometers, press gauges, dimensional gauges etc.
Why Testing equipment register require?
Measurement accuracy
If you don’t have any list of equipment, there may be chance to overdue for calibration, due to improper records. Every product specification depends on the accurate measurements, and the register provides accurate records to identify the details that avoid any mistakes.
Standards compliance
If you follow any international standards that you have to maintain document that provide you proper details of testing equipment. The register may help you to maintain the records as per standards requirements. Usually, ISO 9001(Control of monitoring & measuring resources), ISO 17025 (Requirements of testing lab), GMP (Accurate instrumentation for pharma, and chemical industries. These standards are expecting to comply their requirements for avoiding any failures.
Cost Reduction
If the quality team consistently monitoring, and calibrate equipment. They can avoid unexpected downtime, decrease the replacement costs, avoid production rework / rejection, as well as extend equipment self-life.
How to prepare register?
At the initial stage of the register for preparation, you have to identify all testing equipment installed at various places. The structural process may help to identify the requirements, and easier to list out the equipment, instrument, and devices that use for the measuring process.
At the second step is to assignment unique number to each equipment that may easier to tracking and manage for calibration. Systematic unique Identification number also helpful to manage internal communication.
In the third step is to understand and defining calibration requirements. Usually, requirements are as per industrial standards, criticality of equipment, how frequently use the equipment in the process, and environmental conditions are key requirements.
Creating structure of the register that makes easier to understand and implementation. The register should be covering all the calibration, QMS, and standards requirements for testing equipment.
Testing equipment register template
In the register, equipment calibration schedule is also including in it. Hence, we have to describe the details of each equipment due date for calibration, assigned date, department of assigned, calibration workload etc.
For the maintain schedule for calibration needs list of tools with calibration due date, categorize it by month wise, identifying critical equipment, and priority for calibration. For the calibration, frequency is also equally important. Hence, accuracy requirements, environmental conditions, instrument wear and tea, usages intensity, and recommendation from manufacturer details are also important for schedule.
Conclusion
To ensure measurement reliability, product consistency, standards’ compliances, and operational efficiency. The testing equipment register is helpful for it.
The register is helpful for:
Traceability, Quality assurance, customer confidence, audit compliance and decrease defects.
To prevents mistakes, and enhance performance of the internal management system, register sheet provides foundational information.