Develop internal audit system in laboratory
The purpose of the process is to establish internal audit system and effectively implement audit system to comply requirements of the quality system in laboratory.
Quality management system establishment and manage is not just important but timely conducting the inspection and audit of the each stages of the quality system, effective internal audit is also needs and compliance of the quality system to identify treats, non compliance in quality management system for laboratory system, when we talk about the laboratory internal quality management system that means all the applicable causes, quality system requirements & documentations are according to ISO 17025, which is international standards laboratory specifications and requirements. And all the documentation, laboratory activities, process and supporting equipment audit are conducted to identify requirements with current system compliance and implementation of internal quality management system for the laboratory.
Development of internal audit system in laboratory needs to identification of the non compliance in existing system, through implementation actions are helps to manage. Internal audit system development and continual implementation responsibility, and assign part of responsibility for the effective audit system is management representative of quality management system in the laboratory, management representative of quality management system can distribute its responsibilities to supporting team for effective & real time implementation but final authorities and responsible is management representative of each activities, documentations and audit in the quality management system in laboratory.
The major factors to develop internal audit system in laboratory is conducting analytically work in the laboratory are taking position according to the quality system and instructions. Quality system implementation is support to continual improvement in quality of product, routine process, documentation and equipment’s operations within the laboratory activities and working environment. In case any non-conformance are identified in the system that should immediate corrective actions are taken at place that helps to manage continual implementation of the internal management system in laboratory to controlling over the quality.
Internal audit notification
Before the planning of the internal audit for quality system in laboratory, internal audit notification / declaration are conducted through aware the peoples of the laboratory to make the preparation for the internal audit in the upcoming days. Internal audit notification is the kind of initial intimation to all the employees to check out all the requirements, pending works & preparation for the internal audit, see below picture for internal audit declaration letter given to all concern department head:

Internal audit declaration letter or notification letter is also called as internal audit circular for the preparation of internal audit which is circulate for invitation of the meeting to discussion on the subjects of the internal audit and quality system requirements, current situation of the system within individual department. In the declaration of the internal audit purpose of acknowledge peoples about the audit purpose of the audit and reference documentation are used for the audit to peoples be attention and sustain the requirements, system in the routine works for maintain quality system as well as improvement individual department.
Internal audit declaration letter is given to all concern department head which is in scope of the internal audit of ISO 17025 – international standards and its requirements, the letter include the invitation for the general meeting which all concern members are participate for the general discussion for ISO 17025 – quality requirements, departmental internal system and its requirements, general processes, equipment, machinery & service which enhancement & conformity of the products are in center of the discussion.
Planning of Internal audit in laboratory
Internal audit planning is covered the requirements of the ISO 17025, which are specifically includes organization structural chart, technical requirements and processes, machine, testing devise, testing equipment calibration, test methods are covered as a scope of the internal audit in laboratory. Human resources department conducted all the training concern or applicable for the ISO 17025, quality controls of the products and process, safety requirements and all the concern documentation, reports and evidences are be a part of the internal audits and planning is conducted the same. See picture below for the internal audit planning sheet for reference:
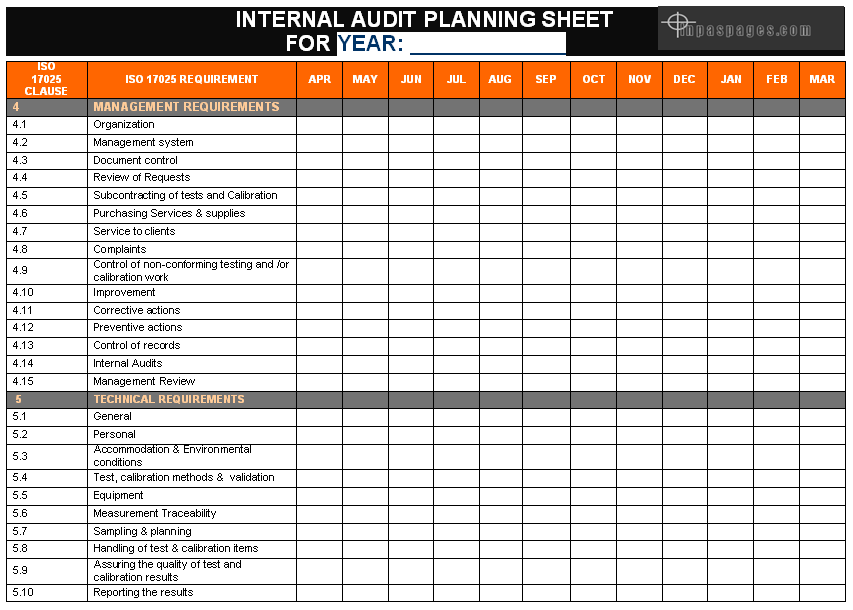
Internal audit planning is conducted an annually shall be prepared by management representative as per international standard, generally planning included scope of audit and previous audits status. Management representative is choose the auditors which are qualified for the internal audit by internal exam or external agency on base of the master list of qualified auditors & previously independently audits. Preparation planning for audit is managed on base of the reviews of relevant processes, previous audit findings and non compliances reports, documents. For the conduct audit checklist helps to investigation, inspect the areas, process and product conformity.
Conducting the internal audit
Internal quality audits conducted with the primary intention of measuring the company’s compliance with:
- Quality System procedures, Work instructions, policy and standard operating procedures etc.
- Business Plans, company objectives and departmental objectives with targets, goals
- Requirements of the quality management systems standards – international standards ISO 17025
- All the government requirements, applicable statutory and regulatory requirements.
- Quality standards, product standards, process standards to meet requirements of the internal quality management system standards
- Safety requirements / Environmental system standard requirements
Internal quality audit conducted to assessing the compliance & implementation of documented procedures & work instructions by qualified auditors, which is not concern with audit department or areas and also audit functional area of the laboratory which contributing specific requirements for the laboratory, auditor verifying the all procedures and individual statements of department peoples as part of audit to ensure that all the concern processes, procedures of standards and instructions are implemented properly. In case any non conformity are raised that should recorded in non conformity report, all the issued Non conformity are corrective actions & preventive actions are submit by auditee.
All audit records should be available with auditors and management representative that helps to know area wise status and finding for implementations of individual department, Auditors team and management representative of quality system are jointly analysis the information collected and audited the areas for the procedures, process and supporting instructions for improvement.
—————————————————————————
DOWNLOAD FORMAT IN WORD | EXCEL | PDF FORMAT
DOWNLOAD INTERNAL AUDIT DECLARATION LETTER
—————————————————————————